|
As the temperature of water increases or decreases, its density decreases. As mentioned earlier, water has its maximum density at 4 degrees Celsius. /beaker-containing-layers-of-fluid-of-different-densities-including-water-and-oil-and-objects-float-93604846-57a7687c3df78cf45916180b.jpg)
The density of water is influenced by several factors, including temperature, pressure, and salinity. Density meters use various techniques, such as oscillation or pressure, to measure the density of water and other liquids accurately. Pycnometers are containers with a known volume that are filled with water and weighed to determine the density. When placed in water, the hydrometer floats, and the level of submersion indicates the water’s density. A hydrometer is a device that consists of a cylindrical glass tube with a weighted bulb at the bottom. There are several methods for measuring the density of water, including hydrometers, pycnometers, and density meters. This is the most commonly used reference point for the density of water. The density of water at 4 degrees Celsius in kg/m³ is 1000 kg/m³. What is the density of water at 4 degree Celsius in kg m3? The density of other substances is often compared to water to determine their relative buoyancy, solubility, and other properties. For example, the density of ice is less than that of liquid water at 4 degrees Celsius, which is why ice floats on water. The density of water at 4 degrees Celsius is often used as a benchmark for the density of other liquids and solids. 
The value of 1000 kg/m³ or 1 g/cm³ is the most commonly used reference point for the density of water at 4 degrees Celsius. These are some of the common units used to express the density of water, and their equivalents in the SI (International System of Units) and the English Engineering system. This is due to the water molecules’ arrangement, which is in a highly ordered and dense structure. At this temperature, water has its maximum density, which means that it occupies the smallest volume for a given mass. This value is often used as a reference point for the density of other substances. The density of water at a temperature of 4 degrees Celsius is 1000 kilograms per cubic meter (kg/m³) or 1 gram per cubic centimeter (g/cm³). In this blog post, we will focus on the density of water at a temperature of 4 degrees Celsius. The density of water changes with temperature, pressure, and salinity. One of the most important properties of water is its density, which is the mass of water per unit volume. Water provides the standard for density at 1 gram/cm 3.Water is an essential element of life, and its properties play a vital role in many scientific and industrial applications. The high heat of vaporization of water makes it an effective coolant for the human body via evaporation of perspiration, extending the range of temperatures in which humans can exist. This provides stability of temperature for land masses surrounded by water, provides stability for the temperature of the human body, makes it an effective cooling agent, and many other benefits. Water has the highest specific heat of any common substance, 1 calorie/gm ☌ = 4.186 J/gm ☌. The water further expands upon freezing, so that water freezes from the top down, and ice floats on water. This decrease in density is only about one part in 10,000 as it cools from 4☌ to 0☌, but this is sufficient to cause the water near freezing to come to the top. 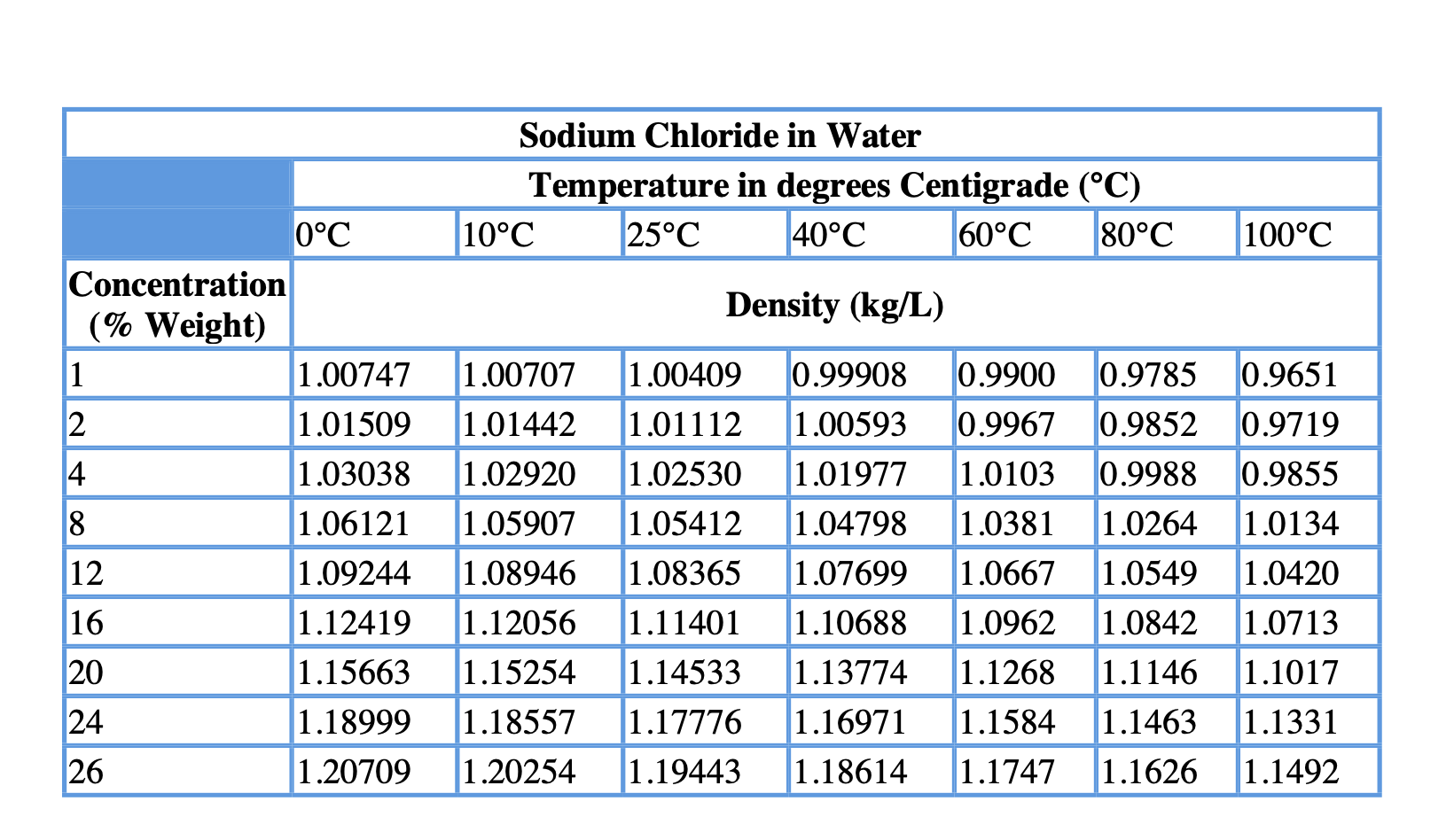
But at about 4☌ it reaches a maximum density and then decreases in density as it approaches the freezing point. Water at ordinary temperatures contracts and increases in density as it is cooled, like most substances. While the hexagonal ice form discussed above is the primary form of ice and is the dominant form from the freezing point at 273K down to about 72 K, 13 different crystalline forms of ice have been idendified according to Debenedetti and Stanley. This hexagonal lattice contains more space than the liquid state. The expansion upon freezing comes from the fact that water crystallizes into an open hexagonal form. The expansion during the phase change may be shown on a PvT surface, and contrasts with the contraction upon freezing of most substances. Then the further expansion as a part of the phase change keeps the ice floating with some 8% of its mass above the surface. The fact that water reaches a maximum density at about 4☌ causes bodies of water to freeze on the top first. The fact that water expands upon freezing causes icebergs to float. The Expansion of Water Upon Freezing The Expansion of Water Upon Freezing
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
